The Cosmetic Regulatory Platform for Innovative Manufacturers & Growing Brands
Ithos is an extension of your R&D and regulatory team.
Create compliant products from start to finish faster, more accurate and easier than ever before.
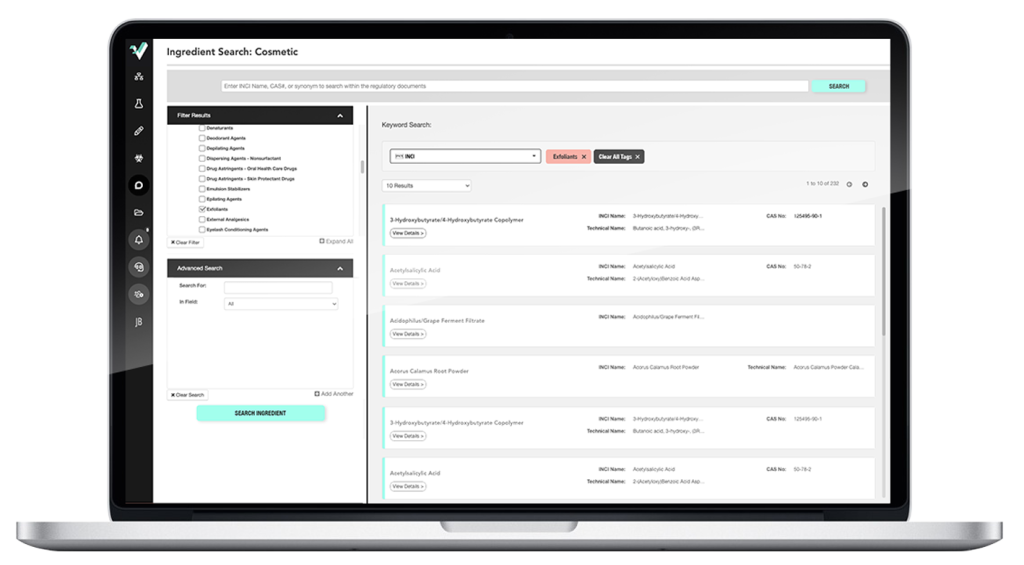
Integrate compliance checks into R&D to launch products faster.
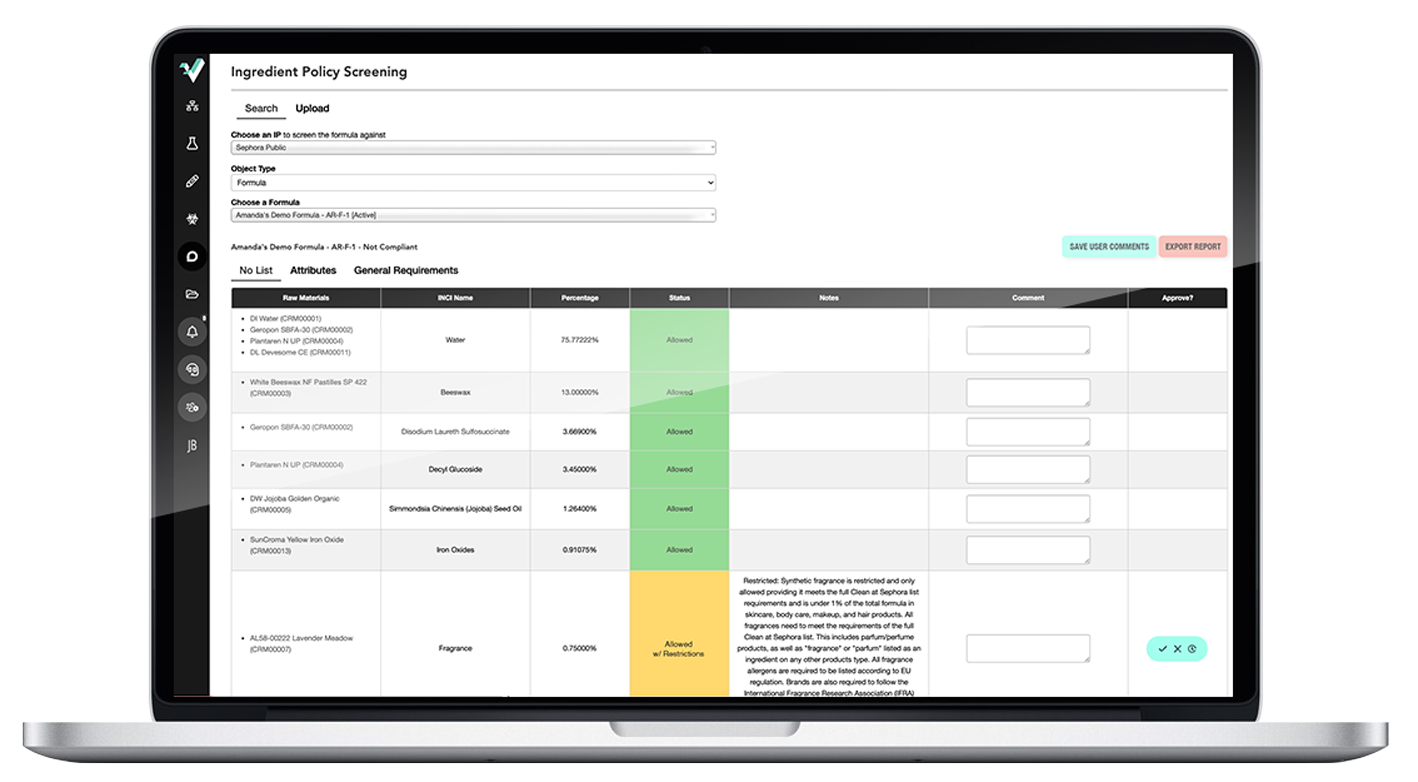
Ithos’s in-house regulatory team has over 75+ years of direct industry expertise.
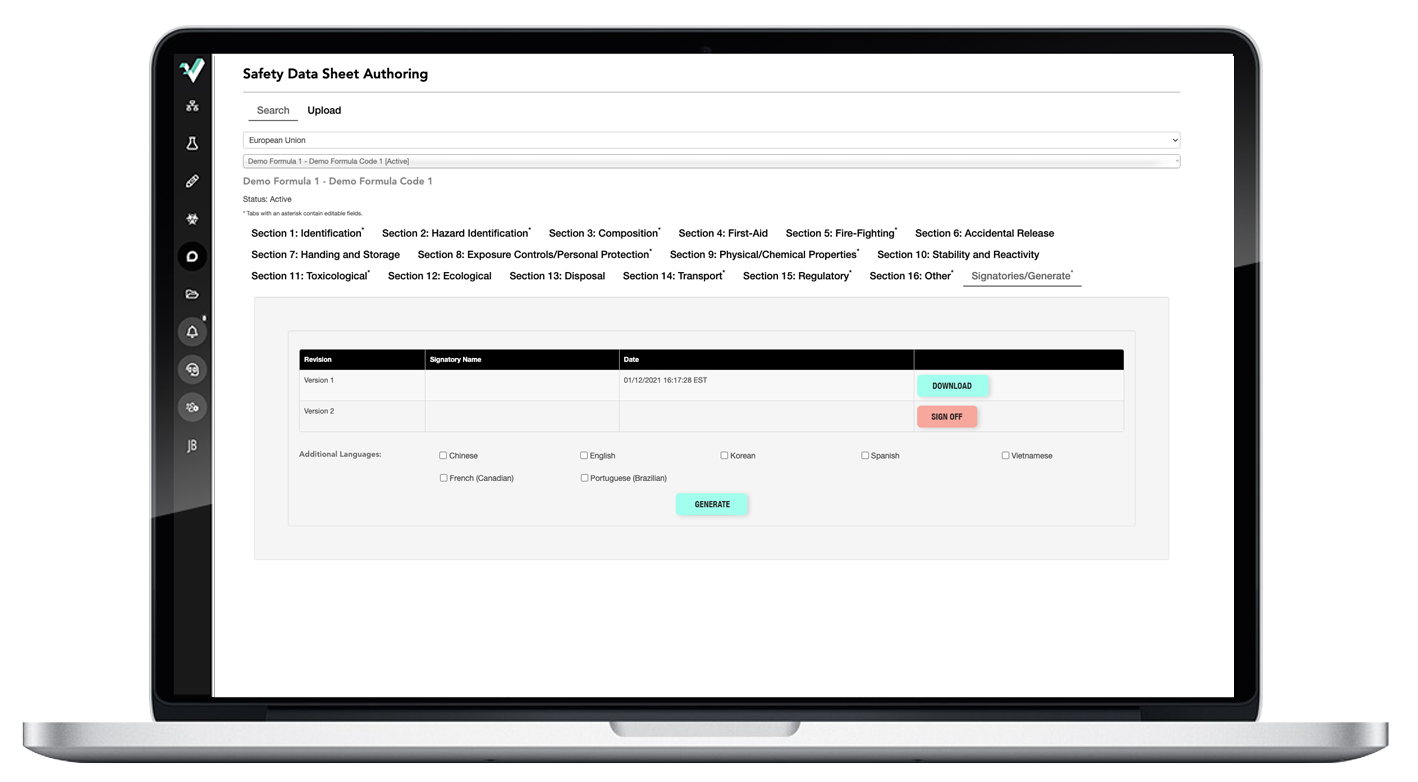
Easy-to-use SDS authoring tool creates WERCS-ready SDSs in minutes.
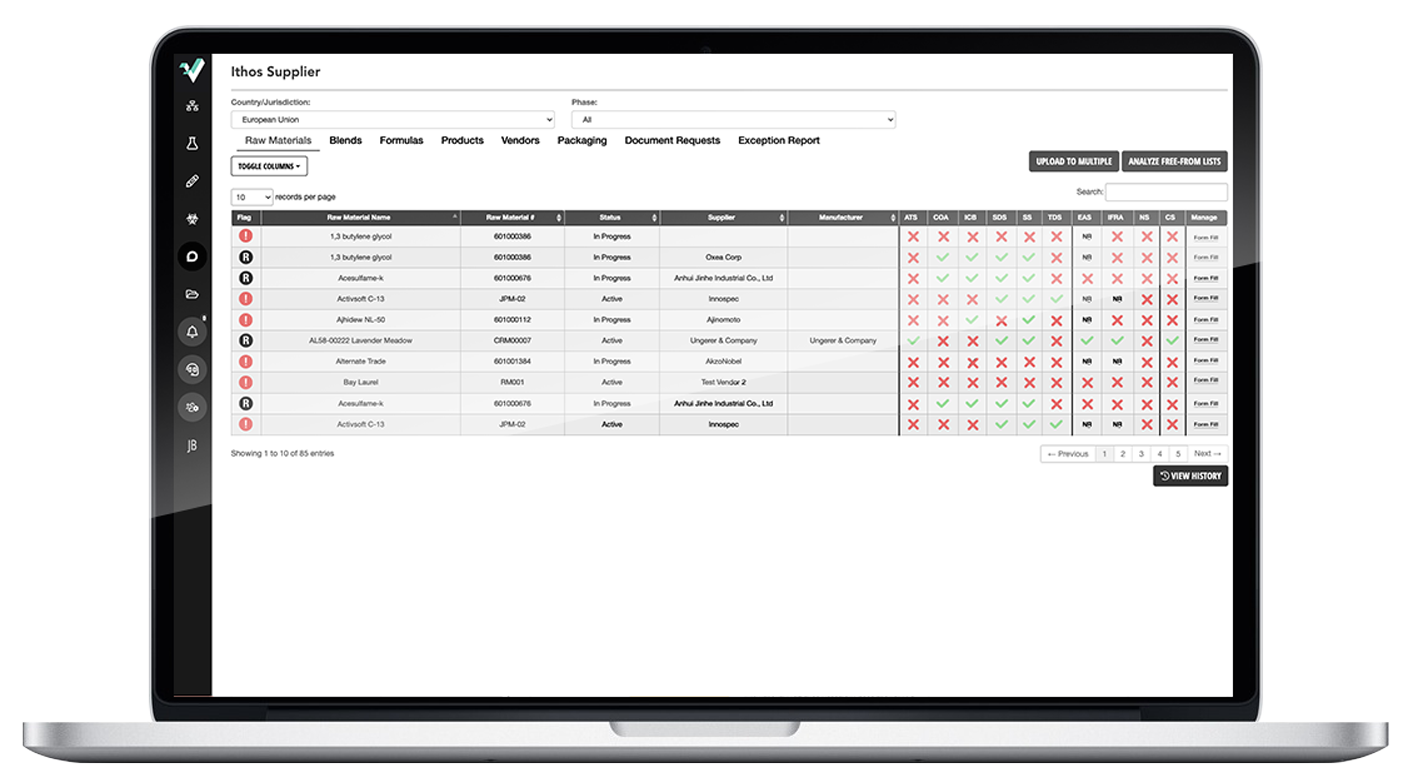
Join the hundreds of others that have taken their companies to the next level with Ithos.




